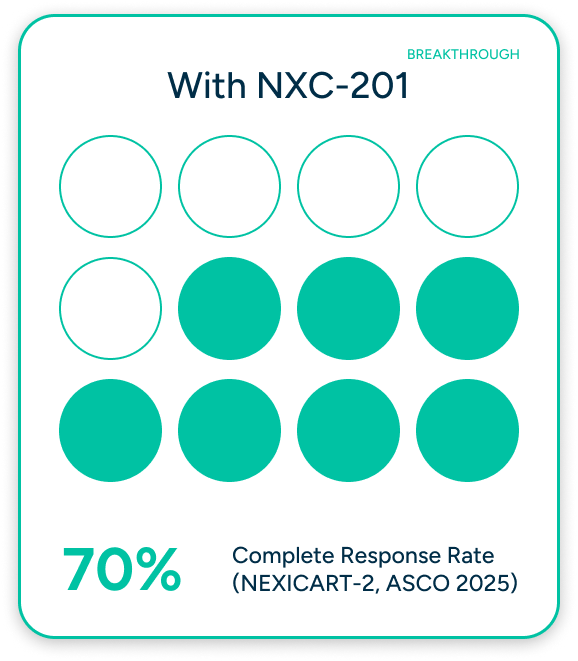
When AL Amyloidosis Comes Back, There's Nowhere to Turn
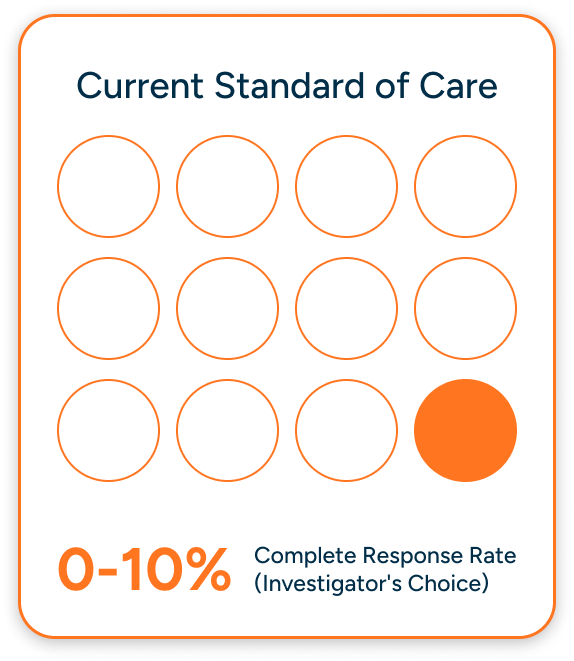
There are no FDA-approved drugs for relapsed/refractory AL amyloidosis. Doctors are forced
to recycle old drug combinations, knowing they'll fail again. Current treatments produce
a devastating 0-10% complete response rate.
For patients with this rare, deadly disease—where toxic proteins attack vital organs, especially the heart—this isn't just a statistic. It's a death sentence.