Changing the Sentence Forever
"Are there any options left for AL Amyloidosis?"
Because of Immix, the answer is finally: "Yes."
We’ve found a breakthrough to change that hopeless sentence. Our mission is simple:
Create medicines that work without destroying the person.
- The Stark Reality
The Current Paradigm is Failing
When AL Amyloidosis Comes Back, There's Nowhere to Turn
There are no FDA-approved drugs for relapsed/refractory AL amyloidosis. Doctors are forced to recycle old drug combinations, knowing they’ll fail again. Current treatments produce a devastating 0-10% complete response rate.
For patients with this rare, deadly disease—where toxic proteins attack vital organs, especially the heart—this isn’t just a statistic. It’s a death sentence.
AL Amyloidosis vs Healthy
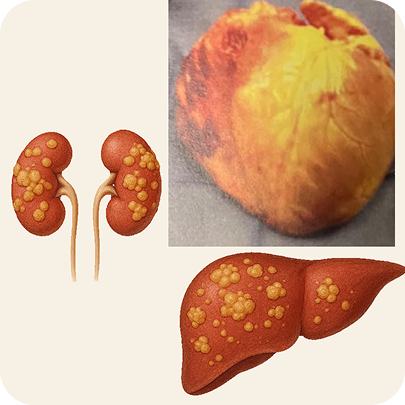
Introducing NXC-201
We Re-Engineer the Immune System
to Fight for You Again
NXC-201 is a revolutionary one-time CAR-T therapy that harnesses the power of your own immune system to eliminate the diseased cells causing AL amyloidosis, targeting the root cause.
Through advanced engineering, we’ve created a treatment that’s both extraordinarily effective and remarkably safe, addressing the critical limitations that have made other therapies impossible for this vulnerable patient population.
Normalizes Disease Within Days
Diseased light chains return to normal levels within a median of just 7 days after treatment, giving your organs the chance to heal.
Single Infusion, Durable Response
Unlike ongoing treatment regimens, NXC-201 requires just one infusion. Your re-engineered immune cells continue working long-term.
Designed for Safety
Zero neurotoxicity. Minimal side effects. The shortest cytokine release syndrome duration of any BCMA CAR-T, critical for protecting vulnerable hearts.
THE SCIENCE BEHIND THE BREAKTHROUGH
Sterically Optimized
Makes the Difference
All BCMA CAR-Ts are not created equal
NXC-201 features three proprietary modifications that deliver “digital” signaling precision while dramatically reducing side effects, particularly critical for AL amyloidosis patients whose primary cause of mortality is heart failure.
- Innovation 1
Optimized CD3ζγ Signaling
Delivers precise “digital” intracellular signaling, reducing non‑specific activation and cytokine release.
- Innovation 2
Enhanced CD8 Hinge
Acts as a “digital filter” preventing inappropriate T-cell activation and further reducing CRS risk.
- Innovation 3
COBRA Binder Technology
Improves plasma cell binding and expression while maintaining flexibility for optimal function.
Extraordinary Results in Clinical Trials
Unprecedented outcomes in one of medicine’s most challenging diseases.
Our U.S. NEXICART-2 registrational trial is delivering results that are transforming the treatment landscape for relapsed/refractory AL amyloidosis.
Current Standard of Care
0%
Complete Response Rate
(Investigator’s Choice)
"An early and deep hematologic response has been found to lead to significantly prolonged survival."
Vaishali Sanchorawala, M.D.
Professor of Hematology and Oncology
Director, Amyloidosis Center at Boston University School of Medicine
0/20
Patients
Normalized disease markers
0 Days
Median Time
To response
0%
Neurotoxicity
Across all patients
4-8x
Shorter CRS
vs. other BMCA CAR-Ts
Beyond AL Amyloidosis: A Platform Built to Transform Multiple Diseases
Immix is laser focused on establishing NXC‑201 as the standard of care for relapsed/refractory AL amyloidosis. Our breakthrough also represents far more than a single‑disease solution.
NXC‑201 targets plasma cells, the antibody factories of the immune system. Success in AL amyloidosis validates our approach and opens the door to treating dozens of immune‑mediated diseases affecting millions of patients worldwide.
Neurology
Rheumatology
Hematology
Vascular & More
$10B+
Total Addressable Market
Across plasma cell-driven immune diseases, with AL amyloidosis alone representing $1.4-2.8 billion in annual sales potential
Beyond AL Amyloidosis: A Platform Built to Transform Multiple Diseases
Immix is laser focused on establishing NXC‑201 as the standard of care for relapsed/refractory AL amyloidosis. Our breakthrough also represents far more than a single‑disease solution.
NXC‑201 targets plasma cells, the antibody factories of the immune system. Success in AL amyloidosis validates our approach and opens the door to treating dozens of immune‑mediated diseases affecting millions of patients worldwide.
Neurology
Rheumatology
Hematology
Vascular & More
$10B+
Total Addressable Market
Across plasma cell-driven immune diseases, with AL amyloidosis alone representing $1.4-2.8 billion in annual sales potential
INVEST IN THE FUTURE
Al Amyloidosis and Other Serious Diseases... Cell Therapy Clinical Trailblazer
Immix Biopharma is a clinical-stage biopharmaceutical company trailblazing cell therapies in AL Amyloidosis and select other serious diseases. Our lead candidate is sterically-optimized BCMA-targeted chimeric antigen receptor T (CAR-T) cell therapy NXC-201. NXC-201 is being evaluated in the U.S. Phase 1b/2a trial NEXICART-2 as well as the ex-U.S. study NEXICART-1.

Want to Learn More?
Latest News
We Are on the Brink of
Turning Despair into Hope
Our progress is accelerating toward commercial launch
Founded Immix Biopharma to change the sentence for patients
2022
- Systematic search launched and concluded to identify breakthrough cell therapies
- Licensed-in global rights intellectual property from Hadassah Israel
2023-2024
- FDA Orphan Drug & FDA Designations secured
- NEXICART‑1 ex‑U.S. trial: 75% complete response rate
- Mentioned in NEJM AL Amyloidosis review
Q1 2025
- Published in Journal of Clinical Oncology
Q2 2025
- American Society of Clinical Oncology (ASCO) Oral Presentation
Q4 2025
- American Society of Hematology (ASH) Oral Presentation
- Onboarded Chief Commercial Officer
2026
- Received FDA Breakthrough Therapy Designation
Coming Next
- BLA submission to FDA
- Commercial Launch
For Patients
If you’ve been diagnosed with relapsed or refractory AL amyloidosis, our clinical trial may offer hope. Learn about eligibility and enrollment.
For Physicians
We’re expanding our network of trial sites. Learn how to refer eligible patients or establish your center as a trial site.
For Investors
Late‑stage biotech with breakthrough clinical data, clear regulatory path, and platform potential worth billions.
Meet the Team
Meet our team of innovators and thinkers at Immix Biopharma, whose dedication and expertise are paving the way for revolutionary treatments in AL Amyloidosis and other serious diseases.
Ilya Rachman is the Chief Executive Officer of Immix Biopharma, Inc. Dr. Rachman is a physician/scientist and former clinical faculty at UCLA. He received both his MD and PhD from the University of Illinois, and his MBA from UCLA Anderson.
Ilya founded a Clinical Research organization that conducted clinical trials of pharmaceutical drugs, and completed several clinical trials as a Principal Investigator building strong relationships in the clinical research industry.
Gabriel Morris is the Chief Financial Officer of Immix Biopharma, Inc. Mr. Morris has been managing partner of Alwaysraise LLC, a life sciences advisory and investment firm based in San Francisco, since founding.
Prior, Mr. Morris was the interim Chief Financial Officer of Zap Surgical Systems, a brain radiosurgery company, where he completed a growth equity financing round. Prior, Mr. Morris led cross-border mergers & acquisitions transactions at Goldman Sachs and other global investment banks for more than a decade, where he participated in greater than $50 billion in completed transactions. Mr. Morris received his B.A. from the Columbia University in the City of New York.
Michael is the Chief Commercial Officer of Immix Biopharma, Inc. Michael has more than 25 years of biopharmaceutical commercialization experience. Prior to joining Immix, Michael was Head of Commercial Operations at Chimerix, Inc., where he led Go-To-Market Strategy and Launch of orphan drug MODEYSO® (dordaviprone), a first-in-disease therapy that was granted accelerated approval in 2025 for H3 K27M-mutant diffuse midline glioma, an ultra-rare and aggressive brain tumor that, prior to MODEYSO®, previously had no FDA-approved therapies. MODEYSO® was granted accelerated approval based on an integrated efficacy analysis of 50 patients, selected from five open-label studies (Jazz Pharmaceuticals acquired Chimerix for $935 million shortly after Chimerix’s New Drug Application for MODEYSO® was accepted and granted Priority Review by the FDA). Prior to Chimerix, Michael was Executive Vice President and Chief Business Officer of Spectrum Pharmaceuticals (SPPI, acquired by Assertio). Prior to Spectrum, Michael held various roles at Amgen in sales and marketing. Michael has been directly involved in driving over 10 successful commercial drug launches throughout his career. Michael holds an MBA from Pepperdine University and a BS from California State University.
Dr. Graydon is a board-certified hematologist-oncologist with over 20 years of experience in clinical development, most recently at Merck & Co. and Johnson & Johnson, where he led new and supplemental new drug applications and biologics license applications for 7 approved drugs including DARZALEX, CARVYKTI, KEYTRUDA, and IMBRUVICA. Dr. Graydon received his MD and PhD from Stanford University and trained at Harvard’s Massachusetts General Hospital.
Mel Davis-Pickett is the Head of CMC Technical Development at Immix Biopharma, Inc. Mel possesses more than 16 years of experience in cell therapy analytical development at Bristol Myers Squibb (BMS), and the Fred Hutchison Institute. At BMS, Mel led analytical development activities for three CAR T programs, including Breyanzi®, from early phase through BLA; co-authored a FDA-approved Justification of Specification for Breyanzi ®’s Module 3, championed stability-indicating method development, and guided portfolio prioritization through cross-functional leadership, risk frameworks, and team mentorship across assay platforms. Prior, Mel was a scientist at the Fred Hutchison Cancer Research Center. Mel received a bachelor’s degree in Cell and Molecular Biology & Chemistry from the Evergreen State College.
We Are on the Brink of
Turning Despair Into Hope
Billions in market potential. Millions of lives changed. This is just the beginning.





